The Medicines and Healthcare products Regulatory Agency (MHRA) in the UK is responsible for ensuring the safety, quality, and efficacy of medicines. One of its critical functions is to oversee medicine recalls and notifications when a product is found to be defective or potentially harmful. Medicine recalls and notifications play a vital role in protecting public health, and the MHRA follows a structured approach to classify and action these recalls based on the risk posed to patients.
The MHRA categorizes medicine recalls into three main classes: National Patient Safety Alert (NatPSA)(equivalent to Class 1 Medicines Recall), Class 2, and Class 3. Each class represents a different level of risk, and the urgency of the action required depends on the severity of the defect. There is also a fourth level, known as Medicines Notification: Caution in use (Class 4).
A table explaining the new Medicines Recall and Notification classifications and the associated defect risk classification can be found on the GOV.UK website.
The Recall Process
Once a medicine defect is identified, the MHRA works with manufacturers, wholesalers and healthcare professionals to assess the risk and determine the appropriate recall classification. The MHRA then issues a Medicines Recall or Notification to inform suppliers, healthcare providers, pharmacies, and the public. For urgent recalls, such as a National Patient Safety Alert (NatPSA)(equivalent to Class 1 Medicines Recall), notifications are sent out immediately to ensure the defective products are removed from shelves and recalled from patients as quickly as possible.
Suppliers, clinics, hospitals, and pharmacies are required to follow the recall instructions, which may include stopping the use of the medicine, returning stock, or advising patients to switch to an alternative treatment. Public communication is a key part of the recall process, ensuring that patients are made aware of any risks and advised on what steps to take if they have already used the affected medicine.
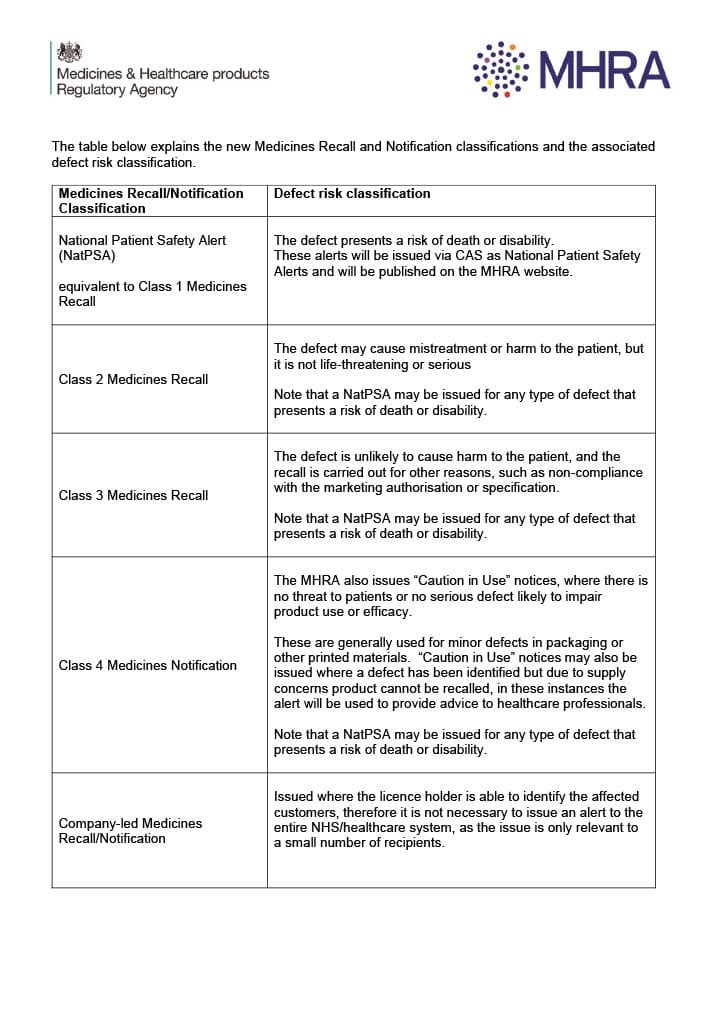
Figure 1: The new Medicines Recall and Notification classifications and the associated defect risk classification. Source: MHRA